Now replace the coefficients with a chemical reaction to get a balanced chemical equation, For example, c1 = 1, and then solve the equation for the coefficients: Now equalize the number of atoms in the reactants to the number of atoms in the products for Carbon ( C ), Hydrogen (H), and Oxygen (O). $$ CH_3 (CH_2)_3 CH_3 + O_2 → CO_2 + H_2O $$Īdding stoichiometric coefficients ( ci)on both sides, To get more familiarity with balancing equations balance the following chemical equations:Įxample 1: Consider the following unbalanced chemical equation: Count the atoms on each side and repeat the steps until you have balanced your chemical equation.Įxamples of chemical equations and balancing:.Now, change the coefficients of one substance.First of all, count the atoms present on each side.Some steps should be followed to balance a chemical equation. How to Balance the Chemical Equations?Ĭhemical equations can be balanced manually. For deep analysis of hydrogen and oxygen in this reaction, you must try out the stoichiometry ratio calculator available online. This example represents a chemical reaction between hydrogen gas and oxygen gas which proceeds to form water. Let's determine the stoichiometry online by an example: Reactants are those elements or substances which we enter in the reaction and products are those elements or substances which are formed as a result of the reaction. To depict reactants and products, it uses chemical formulas. What is Meant by a Chemical Equation?Īn equation for a chemical reaction is a symbolic representation of the reaction. The moment you leave, you will surely be satisfied by our online tool. It is understandable whether you are a beginner or an expert. Our online balancing chemical equations calculator is always here for you to make your problems regarding chemical equations easy.Ĭhemical equation solver makes the process easy and simple, guiding you in steps. Step 3.A balance equation solver is a tool that provides the steps of solving chemical equations for you in seconds. This gives us total changes of +2 and -2. You need 2 atoms of #"H"# for every 1 atom of #"Zn"#. Equalize the changes in oxidation numberĮach #"Zn"# atom has lost two electrons, and each #"H"# atom has gained one electron. Identify the atoms that change oxidation number Here's how the oxidation number method works for a very simple equation that you could probably balance in your head. The general idea is that electrons are transferred between charged atoms. The oxidation number method is a way of keeping track of electrons when balancing redox equations. None of it will happen if you don't get the oxidation number of every player in the reaction. It's just one process and one method with variations. Sometimes one method is more convenient than the other method. In both methods, you must know what the oxidation numbers are and what they become during the reaction. Then you add the two half reactions together and balance the rest of the atoms. Then you multiply them by small whole numbers to make the loss and gain of electrons equal. In the half-reaction method, you determine the oxidation numbers and write two half-reactions. You are making the total loss of electrons equal to the total gain of electrons. Then you multiply the atoms that have changed by small whole numbers. In the oxidation number method, you determine the oxidation numbers of all atoms. Then you balance by making the electron loss equal the electron gain. The only sure-fire way to balance a redox equation is to recognize the oxidation part and the reduction part. They are just different ways of keeping track of the electrons transferred during the reaction. There's no real difference between the oxidation number method and the half-reaction method. Now try to balance the equations in the link below (answers included). 
Place these numbers as coefficients in front of the formulas containing those atoms.ĢHNO₃ + 3H₃AsO₃(aq) → 2NO(g) + 3H₃AsO₄(aq) + H₂O(l)īalance all remaining atoms other than H and O. This gives us total changes of -6 and +6. We need 2 atoms of N for every 3 atoms of As. 
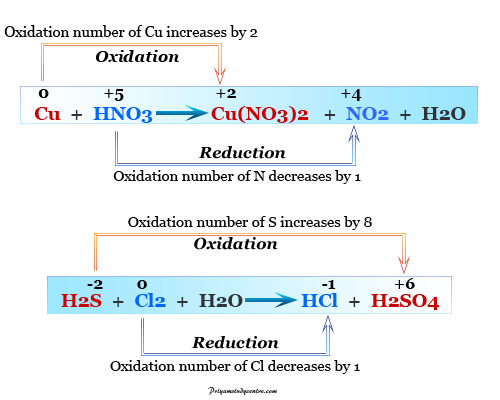
Make the total increase in oxidation number equal to the total decrease in oxidation number. Right hand side: N = +2 O = -2 H = +1 As = +5ĭetermine the change in oxidation number for each atom that changes. Left hand side: H= +1 N= +5 O = -2 As = +3 Identify the oxidation number of every atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
